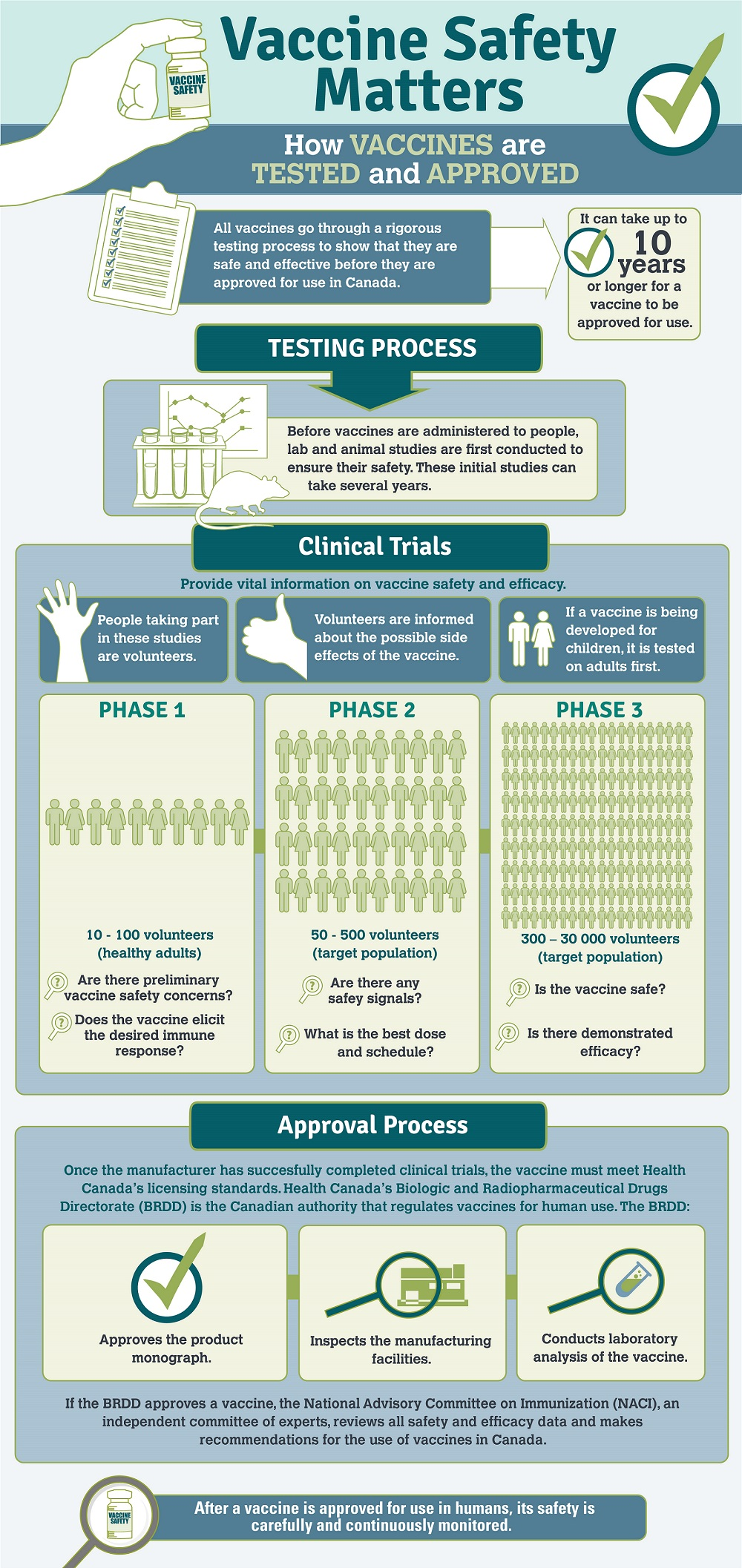
Vaccine safety has become a pivotal topic in public health discussions, especially amid rising vaccine misinformation and controversies leading to significant impacts on health policies. The recent resignation of Dr. Peter Marks from the FDA highlights the tension between political pressures and scientific integrity in evaluating vaccine efficacy, particularly concerning the COVID-19 vaccine approval process. Marks, criticized for not aligning with anti-vaccine narratives promoted by figures like Robert F. Kennedy Jr., expressed deep concerns about the influence of such misinformation on public perception. His departure adds strain to an already beleaguered agency, grappling with challenges from within and external scrutiny regarding its handling of vaccine safety. As debates rage on, the focus remains on ensuring that accurate information prevails over unfounded fears, emphasizing the importance of trust in vaccines as a crucial element of public health.
The discussion surrounding inoculation safety has gained intensified scrutiny, particularly with significant events like the upheaval at the FDA drawing attention to health governance issues. Terms such as vaccine trustworthiness and immunization efficacy have become central to conversations, especially with figures like Robert F. Kennedy Jr. amplifying skepticism about vaccine protocols. The recent shifts within the FDA, marked by the resignation of top officials such as Dr. Peter Marks, underline the challenges agencies face in maintaining public confidence amid a backdrop of political controversy regarding vaccination practices. The ongoing debates reflect a larger struggle against misinformation, calling for clear and credible information to safeguard public health. Ensuring that the benefits of vaccination are communicated effectively is crucial in dispelling myths and reinforcing the foundational trust in vaccination programs.
Understanding Vaccine Safety in the Era of Misinformation
In recent years, vaccine safety has become a contentious topic in public discourse, exacerbated by the rise of vaccine misinformation. The resignation of Dr. Peter Marks from the FDA highlights the deep-seated issues within vaccine regulation and public trust. Marks voiced his concerns about the negative influence of misinformation on vital vaccine policies, emphasizing that truth and transparency must guide health decisions. His willingness to engage with critics like Robert F. Kennedy Jr. demonstrates an openness to dialogue, despite the challenges posed by entrenched anti-vaccine narratives.
The consequences of vaccine misinformation can be dire, as seen in the resurgence of preventable diseases like measles. Public health experts stress the importance of maintaining confidence in vaccines, which have historically saved millions of lives. Marks’ departure from the FDA raises questions about how future vaccine safety assessments will be managed amid heightened scrutiny and skepticism from various fronts, including political figures who may not prioritize scientific evidence over public narrative.
Impact of FDA Leadership Changes on Vaccine Approval Processes
The resignation of Dr. Peter Marks marks a significant shift in the leadership of the FDA during a crucial time for vaccine approval processes. As the architect behind Operation Warp Speed, Marks played a pivotal role in expediting the development of COVID-19 vaccines. His departure could lead to potential disruptions in the approval timeline for future vaccines, as the FDA may adopt a more cautious approach under new leadership, especially with the recent emphasis on vaccine safety amid controversies surrounding figures like Robert F. Kennedy Jr.
The new acting commissioner’s stance on vaccine safety will also likely impact public perceptions and trust in the FDA. The scrutiny surrounding the agency has intensified, especially after marks criticized the influence of misinformation on FDA policies. This situation underscores the critical need for transparent communication from health authorities to counteract the narrative propagated by anti-vaccine advocates. The FDA must pivot strategically to restore trust and ensure the public that thorough scientific evaluation remains at the forefront of vaccine approval processes.
Analyzing the Repercussions of the FDA Vaccine Controversy
The recent controversy surrounding the FDA and vaccine policies has sparked a wave of public reactions and discourse. Dr. Peter Marks’ resignation is viewed as a symbol of the challenges facing the FDA amid political pressures and misinformation campaigns. With prominent figures like Robert F. Kennedy Jr. criticizing vaccine practices, the agency is now under immense scrutiny to demonstrate its commitment to scientific integrity and public health. Experts argue that this turmoil could significantly affect the agency’s ability to enact important health policies.
Moreover, the upheaval within the FDA may have lasting impacts on vaccine uptake and public confidence. As misinformation proliferates, the FDA must devise strategic communication plans that clearly articulate the importance of vaccines in combating preventable diseases. Engaging with communities and responding to their concerns is crucial in reversing the detrimental effects of misinformation and rebuilding public trust in vaccine safety and efficacy.
The Role of Historical Evidence in Supporting Vaccine Safety
Historically, vaccines have played a crucial role in mitigating infectious diseases, dating back centuries. This legacy was echoed in Dr. Marks’ resignation letter, where he reminded readers of the foundational role vaccines have had in improving public health. Scientific evidence overwhelmingly supports the efficacy and safety of vaccines, making the rise of vaccine skepticism all the more alarming. The ongoing measles outbreak serves as a case study in the consequences of eroding trust in scientific consensus, underscoring the need to continuously advocate for vaccination.
As the FDA navigates the complexities of modern vaccine approval processes, it must leverage historical data to combat misinformation. Public health officials need to highlight past successes in vaccination campaigns, showcasing how collective immunity has saved countless lives. Engaging historical narratives can serve as powerful tools in reinforcing the significance of vaccines and countering modern-day skepticism, particularly in light of ongoing debates influenced by contemporary anti-vaccine rhetoric.
Navigating Regulatory Challenges Amidst Vaccine Misinformation
The regulatory landscape surrounding vaccines is evolving, particularly in response to the increasing levels of misinformation and the controversies involving key officials. The resignation of Dr. Marks brings to light significant regulatory challenges that the FDA must navigate. Recent discussions surrounding vaccine safety and responses to figures like Robert F. Kennedy Jr. exemplify the complexity of maintaining scientific rigor while combating pervasive misinformation that influences public opinion.
In addressing these challenges, the FDA must not only uphold stringent scientific evaluations but also actively engage with the community to dispel myths and clarify vaccine safety. This proactive approach will be essential in ensuring that the public remains informed and confident in vaccination programs. Adapting regulatory frameworks to better address public concerns and misinformation will be paramount as health authorities seek to support vaccine safety in the face of evolving societal attitudes.
Public Health Implications of Misinformation on Vaccines
Misinformation surrounding vaccines has profound public health implications that extend beyond individual choice. The backlash against vaccine safety, partly fueled by influential figures such as Robert F. Kennedy Jr., threatens the hard-won gains made in controlling diseases through vaccination. The FDA’s responsibility is to uphold public health standards against this misinformation wave, ensuring that discussions about vaccine safety are grounded in scientific evidence rather than sensationalized rhetoric.
The public health community must prioritize education and outreach to counter the adverse effects of vaccine misinformation. Collaborative efforts among agencies can foster a culture of trust and transparency, essential for overcoming the skepticism that jeopardizes population health. By reinforcing scientifically-backed facts and engaging directly with at-risk communities, public health initiatives can mitigate the negative impacts of misinformation and promote higher vaccination rates.
The Future of Vaccine Policies After Dr. Marks’ Resignation
With Dr. Peter Marks’ resignation, the future direction of vaccine policies within the FDA is uncertain. Marks was a strong advocate for swift and evidence-based vaccine approvals, especially during the COVID-19 pandemic. His departure may lead the FDA to take a more cautious approach, thus impacting the timeline for future approvals. The need for a clear strategy on vaccine safety and communication will be crucial for the incoming leadership to maintain trust among the public and healthcare professionals.
As vaccine-related controversies unfold, the FDA must reaffirm its commitment to transparency and robust scientific evaluation. The agency’s ability to navigate these challenges will shape the future landscape of vaccination in the U.S. By prioritizing dialogue with communities and addressing misinformation directly, the FDA can ensure that public health remains a priority despite rising skepticism and questions surrounding vaccine safety.
Reinforcing Public Confidence in Vaccines After Controversy
The controversies surrounding vaccine approval processes have significantly impacted public confidence. Following the resignation of Dr. Peter Marks, the FDA faces the daunting task of restoring trust in its vaccine safety protocols. Strategies to reinforce public confidence must include proactive communication that addresses concerns and misinformation directly, particularly as narratives shaped by figures like Robert F. Kennedy Jr. continue to gain traction.
To rebuild this trust, the FDA could enhance community outreach programs emphasizing the value of vaccines supported by scientific research. Engaging healthcare providers as trusted sources of information can also play a pivotal role in dispelling myths and misinformation about vaccine safety. By fostering partnerships with public health advocates and utilizing educational resources, the agency can work towards mending the rift in public trust and promoting vaccination as a cornerstone of public health.
Vaccine Approval Process: Striking a Balance Between Safety and Urgency
The vaccine approval process is characterized by the need to balance safety with the urgency of public health emergencies. The resignation of key figures at the FDA, including Dr. Marks, reflects the ongoing tension between hastening vaccine development in response to crises like the COVID-19 pandemic and maintaining stringent safety protocols. This tension is emblematic of the broader challenges faced within the agency as it seeks to reassure the public while countering vaccine misinformation.
Future discussions on vaccine policies must consider how to streamline approvals without sacrificing the robustness of safety evaluations. Transparent communication about the processes involved in vaccine safety evaluations will be key in addressing public concerns. By prioritizing these efforts, the FDA can assure the public that despite the urgency of health crises, the integrity of vaccine safety will remain uncompromised.
Frequently Asked Questions
What are the impacts of vaccine misinformation on public perception of vaccine safety?
Vaccine misinformation severely impacts public perception of vaccine safety by eroding trust in health authorities and established scientific consensus. High-profile figures, like Robert F. Kennedy Jr., have propagated anti-vaccine narratives that contradict decades of research supporting vaccine efficacy and safety. This misinformation can lead to decreased vaccination rates, resulting in outbreaks of preventable diseases, like the ongoing measles outbreak highlighted in recent reports.
How does the FDA address vaccine safety amid controversies such as the recent Peter Marks resignation?
The FDA has a robust system in place for evaluating vaccine safety, even amidst controversies like the resignation of Dr. Peter Marks, who criticized the undue influence of misinformation. The FDA’s protocols are designed to ensure rigorous review processes for vaccines, including COVID-19 vaccine approvals, which have been backed by substantial clinical data. The resignation highlights the challenges posed by misinformation but does not diminish the FDA’s commitment to evidence-based decisions.
What criticism has the FDA faced regarding vaccine safety oversight, especially in the context of political controversies?
The FDA has faced criticism regarding its oversight of vaccine safety, particularly in light of political pressures from figures like Robert F. Kennedy Jr., who has challenged the agency’s integrity. Dr. Marks’ resignation letter expressed concern about how misinformation compromises public trust and the agency’s ability to promote science-based conclusions about vaccine safety. Such tensions underscore the importance of maintaining scientific integrity in public health matters.
What role do health officials play in combating vaccine misinformation and ensuring vaccine safety?
Health officials play a crucial role in combating vaccine misinformation by providing accurate, transparent information about vaccine safety and addressing public concerns. The recent controversy surrounding Dr. Peter Marks’ resignation illustrates the challenges faced when misinformation influences policy. Continued advocacy and education from health officials are vital to restoring trust and ensuring successful vaccine uptake, particularly for the COVID-19 vaccine and childhood vaccinations.
How does historical evidence of vaccinations support ongoing discussions about vaccine safety?
Historical evidence demonstrating the benefits of vaccines is vital to discussions on vaccine safety. Figures like George Washington supported vaccination efforts, underscoring a long-standing tradition of vaccination as a key strategy in public health. Dr. Marks cited these historical precedents and ongoing outbreaks, like measles, to emphasize that skepticism towards vaccines can lead to severe public health consequences, reinforcing the critical nature of vaccine safety.
| Key Point | Details |
|---|---|
| FDA Vaccine Official Resigns | Dr. Peter Marks announced his resignation in a letter criticizing misinformation about vaccine safety promoted by U.S. health officials. |
| Criticism of Health Officials | Marks accused the Secretary of Health of seeking validation for misinformation rather than truth and transparency regarding vaccine safety. |
| Response to Misinformation | Marks was open to addressing concerns raised about vaccine safety but found it unfeasible due to the prevailing misinformation. |
| Impact of Resignation | His exit raises concerns about the commitment to science and transparency in public health policymaking. |
| Ongoing Vaccine Safety Debate | Despite overwhelming evidence supporting vaccine safety, some officials, like Robert F. Kennedy Jr., have pushed for scrutiny, potentially leading to public skepticism. |
Summary
Vaccine safety is a critical public health issue that continues to be influenced by misinformation and personal beliefs. The recent resignation of Dr. Peter Marks from the FDA underscores the serious challenges facing vaccine advocates. His criticism aimed at the manipulation of facts about vaccines points to an urgent need for a unified effort to reinforce trust in vaccines. Continued misinformation poses a risk not just to public confidence but to the overall safety and efficacy of vaccination programs that have successfully protected populations from preventable diseases.